 Abstract
Abstract
Background: Prevention of superficial surgical wound infections from drug-resistant bacteria such as methicillin resistant Staphylococcus aureus (MRSA) currently present major health care challenges. The majority of surgical site infections (SSI) are believed to be caused by airborne transmission of bacteria alighting onto the wound during surgical procedures. We have previously shown that far-ultraviolet C light in the wavelength range of 207-222 nm is significantly harmful to bacteria, but without damaging mammalian cells and tissues. It is important that the lamp be fitted with a filter to remove light emitted at wavelengths longer than 230 nm which are harmful.
Aims: Using a hairless mouse model of infection of superficial wounds, here we tested the hypothesis that 222-nm light kills MRSA alighting onto a superficial skin incisions as efficiently as typical germicidal light (254 nm), but without inducing skin damage.
Methods: To simulate the scenario wherein incisions are infected during surgical procedures as pathogens in the room alight on a wound, MRSA was spread on a defined area of the mouse dorsal skin; the infected skin was then exposed to UVC light (222 nm or 254 nm) followed by a superficial incision within the defined area, which was immediately sutured. Two and seven days post procedure, bactericidal efficacy was measured as MRSA colony formation unit (CFU) per gram of harvested skin whereas fixed samples were used to assess skin damage measured in terms of epidermal thickness and DNA photodamage.
Results: In the circumstance of superficial incisions infected with bacteria alighting onto the wound, 222-nm light showed the same bactericidal properties of 254-nm light but without the associated skin damage.
Conclusions: Being safe for patient and hospital staff, our results suggested that far-UVC light (222 nm) might be a convenient approach to prevent transmission of drug-resistant infectious agents in the clinical setting.
Conflict of interest statement
Figures
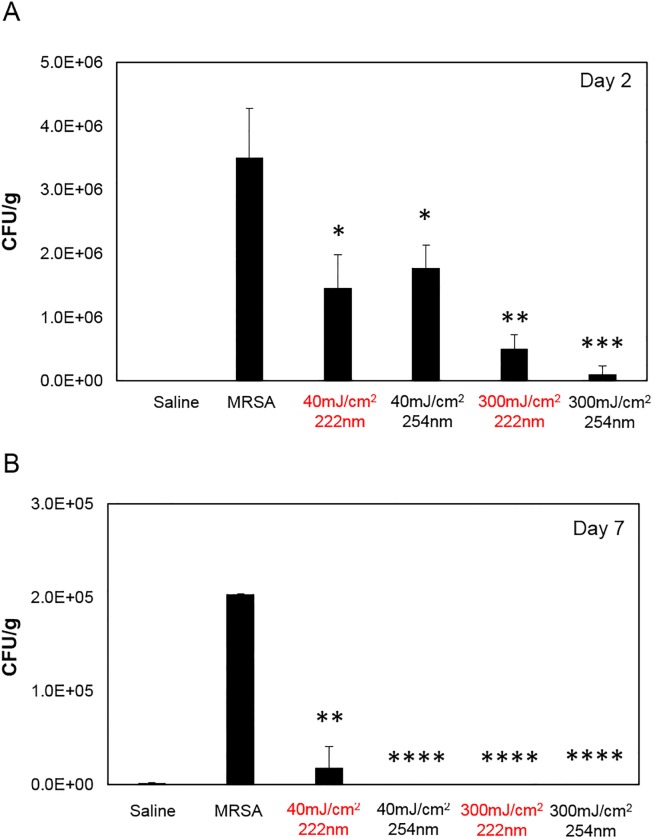
Fig 1. Bactericidal efficacy of UVC light of superficial wounds infected with MRSA.
CFU/g of mouse skin where MRSA was spread before exposure to 40 or 300 mJ/cm2 delivered by the 222- or 254-nm light, and followed by a superficial incision. Negative controls were obtained from skin incisions that were uninfected by applying saline prior to exposure to either UV lamp (Saline), whereas skin incisions that were infected but not exposed to the UV light (MRSA) represented the positive controls. Tissues were harvested at A) day 2 or B) day 7 after exposure. *p< 0.05, **p< 0.01, ***p< 0.005, ****p< 0.0001 compared to positive controls (MRSA).

Fig 2. Epidermal thickness of mice skin infected with MRSA and exposed to UVC light.
A) Representative cross-sectional images of hematoxylin and eosin-stained mouse dorsal skin comparing the epidermal thickness two or seven days after exposure to 40 or 300 mJ/cm2 from the 222-nm or 254-nm light to that of skin incisions that were uninfected by applying saline prior to exposure to either UV lamp (Saline). Skin incisions that were infected but not exposed to the UV light (MRSA) represented the positive controls. B) Quantification of epidermal thickness; values represent the average ± SD measured in nine randomly selected field of view per mouse. *p< 0.05, **p< 0.005 compared to positive controls (MRSA).

Fig 3. DNA photodamage in mice skin infected with MRSA and exposed to UVC light.
